

In recent years there have been introduced spiral periodic charts as a revolutionary new presentation of relationships between the elements similar to the structure of spiral galaxies. But in fact Gustavus Detlef Heinrichs (b1836, d1923), professor at the University of Iowa, published in 1866 a paper claiming that the properties of elements are funcions of their atomic weights. The following year he published a book with a a figure much like a spiral periodic chart.
One chart representative of the useful patterns is that of Theodor Benfey (shown below with his permission)
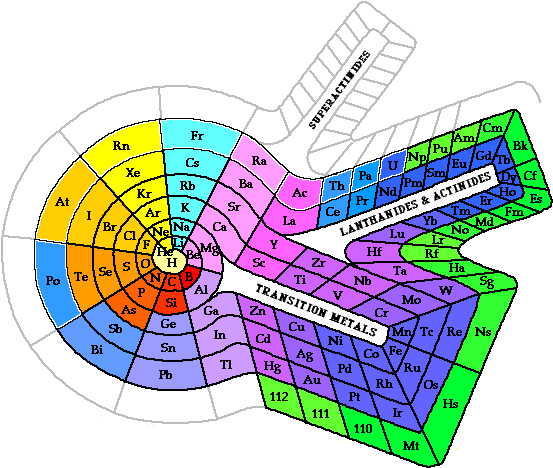
I developed the spiral table while I was editor 1963-88 of the ACS chemical education magazine Chemistry.... It is hard to see anything very periodic in the usual tables hanging in classrooms so I developed this one which shows a beautiful step-wise periodicity. After two periods of 8 there are two of 18, then two of 32 etc. When a forthcoming article in Chemistry talked about a particular set of elements, we showed and highlighted that section of "our" table. Then the chemical instrument company Instruments for Research and Industry (I2R) discovered the table and used it in the September page of its calendar. It has used the table in several years since, including this year when it merged with the Glas-Col company.
The spiral table is making its way into the mainstream. It has been used in two general chemistry textbooks and on the cover of Geoff Rayner-Canham's Descriptive Inorganic Chemistry. It is reproduced in William Brock's Fontana History of Chemistry (in the US: Norton History of Chemistry), and in Germany, in a Max Planck research institute, it is the floor plan under the swings of a Foucault pendulum.
Ted (Theodor) Benfey,
Emeritus Prof. of Chemistry and History of Science
Guilford College
Editor-at-Large, Chemical Heritage


| introduction | Greeks | alchemy | Lavoisier | Dalton | Berzelius | molecules | spectra | electron | radiation | Bohr | isotopes | synthesis | ↑ |
| Theodor Benfey's chart | to site menu | Discovery and Naming of Chemical Elements |
chemistry | physics | |||||||||
| created 26 October 2006 latest revision 4 May 2010 |
by D Trapp | ||||||||||||