

Alchemy began as an Arab mixture of Egyptian arts of dyeing, painting, glass making, pyrotechnics, medical drugs, mining, and metallurgy with the theories of the Greeks (mostly Aristotle's) to explain changes in color and appearance of materials.
One of the main goals of alchemists was to learn the proper proportions of ingredients and processes to cause one element to undergo transmutation to another element.
The earliest known Alchemists were Zosimus and Synesius (perhaps of the 3rd Century A.D.). Writings attributed to them described practical laboratory operations, but used obscure symbols, probably to maintain Alchemy secrets.
Aristotle's four elements are a single materia prima with qualities of hot/cold and moist/dry. Other perceived properties such as smell, taste, and color are actually products of the primary qualities. One element could be changed to another by changing these attributes.
Over 200 alchemy books are attributed to Jabir ibn Hayyan (722 to 815 A.D.).
Emphasis was placed on first hand knowledge of materials. For he who performs not practical work nor makes experiments will never attain to the least degree of mastery. ...Do thou experiment so that thou mayest acquire knowledge.
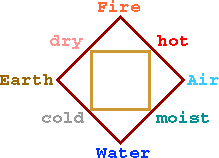
The properties of hot/cold and moist/dry can be separated from materials and combined in definite proportions to form desired materials. Thus the alchemist's job, according to Jabir, is to determine the proper proportions for preparing desired products.
If metals are deprived of their attributes, they can be reduced to Materia Prima, then, for example, given the attributes of gold.
Jabir added to the properties hot/cold and moist/dry of the Greek four elements, the principles of Sulfur and Mercury. Minerals are generated from exhalations in the earth in a two step process. Dry exhalations first produce Sulfur and moist exhalations produce Mercury. Finally, combinations of Sulfur and Mercury form various minerals.
Abu Bakr Muhammad ibn Zakariya al-Razi, a Muslim called Rhazes (860 to 925 A.D.) was the first of a series of physicians that were the chief contributors to chemistry for the next 500 years. While most of his ideas were similar to Jabir, he discovered caustic al-qili.
Abu 'Ali al Husain ibn Abdallah ibn Sena called Avicenna in Europe (980 to 1037) was the greatest physician of Islam. His Canon
of medicine became the standard reference for the next six centuries.
His
De Mineralibusgave a clear account of accepted theories. He classified mercury with the metals, writingit resembles the essential constituent element of all fusible bodies because they all convert to mercury on fusion.He suggested transmutations are not true change of elements, but produce excellent imitations.
Geber (13th Century Spanish adaptation of Jabir) in the Summa Perfectionis
describes formulas to give health, eternal youth, riches, and power. Geber describes the qualities of gold, silver, lead, tin, copper, and iron and explains the sulfur-mercury theory.
The need for correct proportions make accurate balances, previously developed by metal assayers, important for alchemy too. Geber describes them, and notes
lead gains weight when calcined because spirit is united with the body.
| SPIRITS (volatized entirely by fire) |
METALS (fusible substances hammering gives noise) |
(BODY or MINERAL (shatter or pulverize when hammered) |
| sulfur arsenic mercury* sal ammoniac camphor |
lead tin gold silver copper iron khar sini* (*or mercury if not a spirit) |
a) some spirit but having form of a body: malachite, lapis lazule, turquoise, mica, etc. b) small amount of spirit: shells, pearls, vitriols, etc. c) almost no spirit: oxyx, dust, aged vitriols, etc. |
| these had links to planets and weekdays |


| introduction | Greeks | ↑ | Lavoisier | Dalton | Berzelius | molecules | spectra | electron | radiation | Bohr | isotopes | synthesis |
| to site menu | Discovery and Naming of Chemical Elements |
chemistry | physics | |||||||||
| created 19 March 2002, additions 6 April latest revision 4 November 2006 |
by D Trapp | |||||||||||